PostScript Acute 15 (Feb 2014)
To view a PDF of this edition click here.
Information included is specific to the use of medicines in the adult setting.
Antiepileptic drugs and brand prescribing
Following a recent MHRA alert on brand prescribing of antiepileptic drugs (AEDs), the Area Drug & Therapeutics Committee issued the following guidance to primary care (for full details see January edition of PostScript):
|
Antiepileptic drug (AED) |
Advice |
|
Phenytoin Carbamazepine Phenobarbital Primidone |
Maintain patient on a specific brand or manufacturer’s preparation. |
|
All other AEDs
|
Patients are not required to be maintained on a specific brand or manufacturer’s preparation, unless the prescriber endorses this on the prescription. |
In light of this, the following is recommended for patients admitted to hospital:
Immediate action on admission:
- Initially prescribe all AEDs generically (to avoid the risk of missed or delayed doses).
- Administer patient’s own AEDs; if not available (or unsuitable for use) use ward stock, irrespective of brand or manufacturer.
Within 48 hours of admission:
- Ensure that epileptic patients prescribed one or more of the following drugs have the brand or manufacturer of their AED endorsed on the Kardex (and ordered from pharmacy if the patient’s own supply cannot be used):
- Phenytoin
- Carbamazepine
- Phenobarbital
- Primidone
- Any other AED drug endorsed with the brand or manufacturer on the Emergency Care Summary (ECS)
- If the specific brand or manufacturer cannot be established or obtained, do not miss or delay doses - use an alternative brand and monitor the patient for signs of uncontrolled epilepsy and drug toxicity (checking AED plasma levels as appropriate).
- Patients on other AEDs do not require brand prescribing.
Please note:
- Pharmacy, where possible, will provide support with the above.
- Brand prescribing is NOT required if the AED is used for the management of neuropathic pain.
Remember…
- Missed doses of antiepileptic drugs (AEDs) can precipitate seizures.
- To avoid this, initially prescribe AEDs generically and review the need for brand prescribing within 48 hours of admission to hospital.
TrakCare discharge prescriptions: What do pharmacists check?
A study1 investigating the perceived causes of prescribing errors by junior doctors highlights a range of factors. Discharge prescriptions are considered particularly problematic, with junior doctors feeling under extreme pressure to complete them quickly so that beds are available for new admissions.
‘That’s probably not brilliant…. but I suppose like stuff like that, I know someone else is going to check it, whether it’s the pharmacist or whether…. I know that somebody will go through it the next morning and confirm….’ - FY1 Doctor
This quote from the study illustrates a growing problem associated with prescribing errors, namely an assumption that errors will always be intercepted, usually by the pharmacist, before they reach the patient. This assumption is incorrect. Pharmacists DO NOT check every medicine prescribed on a Kardex for every patient, and DO NOT always have full patient information available to them when checking a patient’s discharge or out-patient prescription. The following reported medication incident illustrates the risk in practice:
Medication incident
- Patient discharged on bumetanide 3mg once a day instead of 3mg twice a day.
- Patient re-admitted some time later with fluid overload.
- Investigation revealed that the prescriber had written the wrong dose in the IDL (the patient was prescribed 3 mg twice a day on the Kardex).
- The pharmacist reviewing the IDL did not have access to the patient’s medical notes or Kardex and was therefore not in a position to identify and correct the error.
In NHSGGC the Immediate Discharge Letter (IDL) must be reviewed by a pharmacist before it can be authorised for dispensing. Where possible, this is done by the clinical pharmacist who has reviewed the patient at ward level with access to their medical notes and Kardex. However, this is not always possible and furthermore not all patients are reviewed by a clinical pharmacist during their admission. In these circumstances a pharmacist, unfamiliar with the patient’s history and without access to their medical notes or Kardex, will review the IDL and check the following:
- The dosage and frequency for each medicine are appropriate for a ‘standard’ patient according to the BNF & patient’s age. This will not take into account other clinical factors such as co-morbidities, renal or hepatic impairment where ‘standard’ doses may not apply.
- Appropriate duration for each medicine from the limited information available in the IDL.
- Potential drug interactions and pharmacological duplications apparent from the information in the IDL.
This level of check is comparable to that provided by community pharmacists on GP prescriptions.
Please note: The NHSGGC Medicines Reconciliation in Hospital Good Practice Guide (Appendix 7) outlines the process for medicines reconciliation at discharge.
Remember…
- Reliance on a second check is not safe practice. Take care to write an accurate and complete IDL before sending to pharmacy.
- The pharmacist does not always have full patient information to identify and correct all prescribing errors.
Gentamicin and vancomycin online calculators
Ensure care with data entry
- Both the gentamicin and vancomycin online calculators require a variety of patient details to be entered to enable an appropriate dose to be recommended.
- Recent Datix reports have highlighted errors with data entry. Examples include; age being entered in the weight field or eGFR or CRP entered in the Creatinine field.
- Users are reminded to double check all entries before accepting and prescribing the dose provided.
For example: The screen shots below demonstrate how a 40 year old weighing 62kg could be significantly underdosed with gentamicin if the age and weight entries were mixed up.
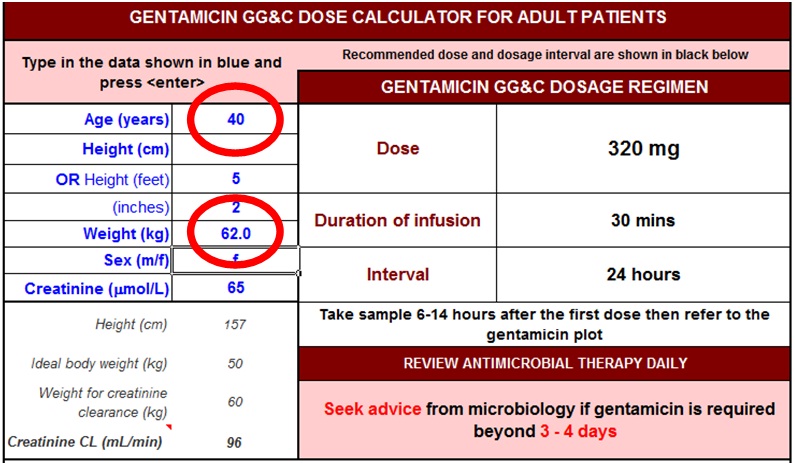
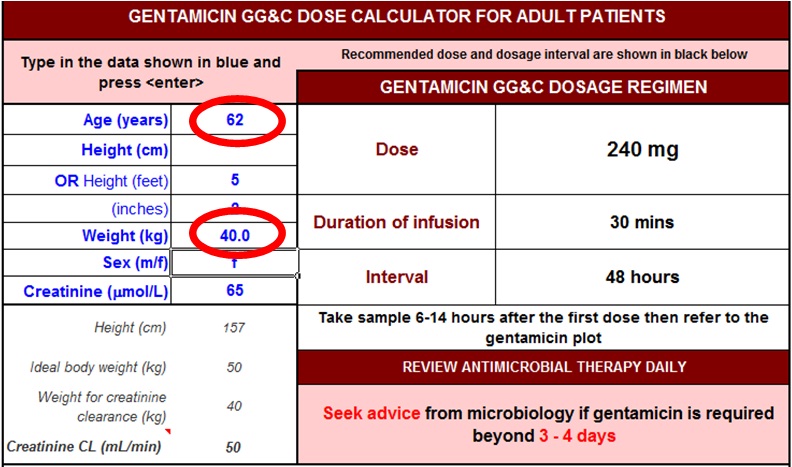
Use ACTUAL body weight
Clarity has been sought regarding the weight to be entered into the calculator. The patient's ACTUAL weight should be entered. The calculator will then adjust the weight if necessary (using the patient's height) to calculate the creatinine clearance.
Emergency Care Summary (ECS): an update
ECS is widely used as a key source of information for medicines reconciliation (Meds Rec), however, it has some limitations which have prompted the following queries and learning points regarding its use:
Use of ECS in different clinical settings
ECS can be used to support medicines reconciliation for:
- Planned and unplanned admissions to hospital.
- Out-patient appointments and continuing care. For example, when a patient with a long term condition is seen periodically by a secondary care specialist or as part of hospice care.
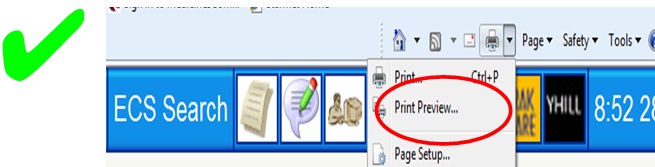
Printing ECS via the Clinical Portal
ECS is most commonly accessed via the clinical portal. There have been reported incidents where a copy of ECS has been printed from portal and the resulting print out had no patient identifier. In at least one case this information was then used to complete medicines reconciliation in the wrong patient.
The problem arises from using the internet browser print options on the portal page.
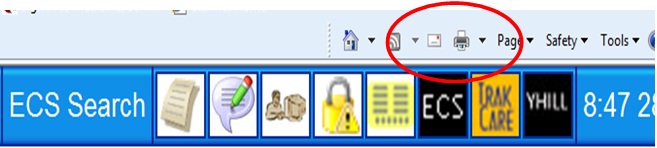
If you select the first ‘Print’ option (see below) then the patient’s name WILL NOT appear on the printed copy.
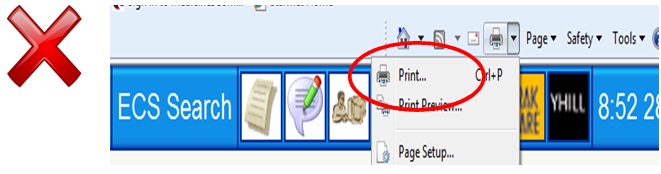
If you select the select the second ‘Print Preview’ option (see below) then the patient’s name WILL appear on the printed copy.
IMPORTANT
Always check the patient’s identity against any information sources being used, as ‘wrong patient’ incidents have occurred even when patient identifiers were on the information source.
Updating the patient record on ECS
ECS is updated daily with any new information in the GP record. If there is no new information then the ECS record remains unchanged. New information may be a change to the medication, allergies or the issuing of a prescription. This explains why the ‘last ECS received’ date shown on an ECS may be some weeks or months previously.
ECS and ‘hospital’ medicines
ECS does not usually include medicines which are prescribed and supplied by hospital/specialist services e.g. clozapine. See PostScript Acute Dec ‘13 for further information.
Guideline News
SIGN Clinical Guidelines:
NICE Clinical Guidelines*:
- Intravenous fluid therapy in adults in hospital (CG 174)
- Prostate cancer (CG 175)
- Head injury (CG 176)
*NICE Guidelines are developed for prescribers in NHS England and Wales and as such may not always follow NHS Scotland prescribing policy e.g. SMC advice. They should always be used in conjunction with relevant NHSGGC Formulary and Clinical Guidelines.
Subscribe to any of the PostScript publications by sending an email to postscript@ggc.scot.nhs.uk
REFERENCES:
1. PROTECT programme. BMJ Qual Saf 2013 22:97-102
